The Michigan Legislature’s newly passed state budget, which protects all existing healthcare funding, was signed into law Oct. 7 by Gov. Whitmer. Public Act 22 of 2025, sponsored by Rep. Ann Bollin (R-Brighton), passed by both …
The MHA brought together the Community Benefit Collaborative members for a daylong kickoff event on Oct. 2 to network and learn from peers across the state conducting community benefit work in chronic disease, behavioral health …
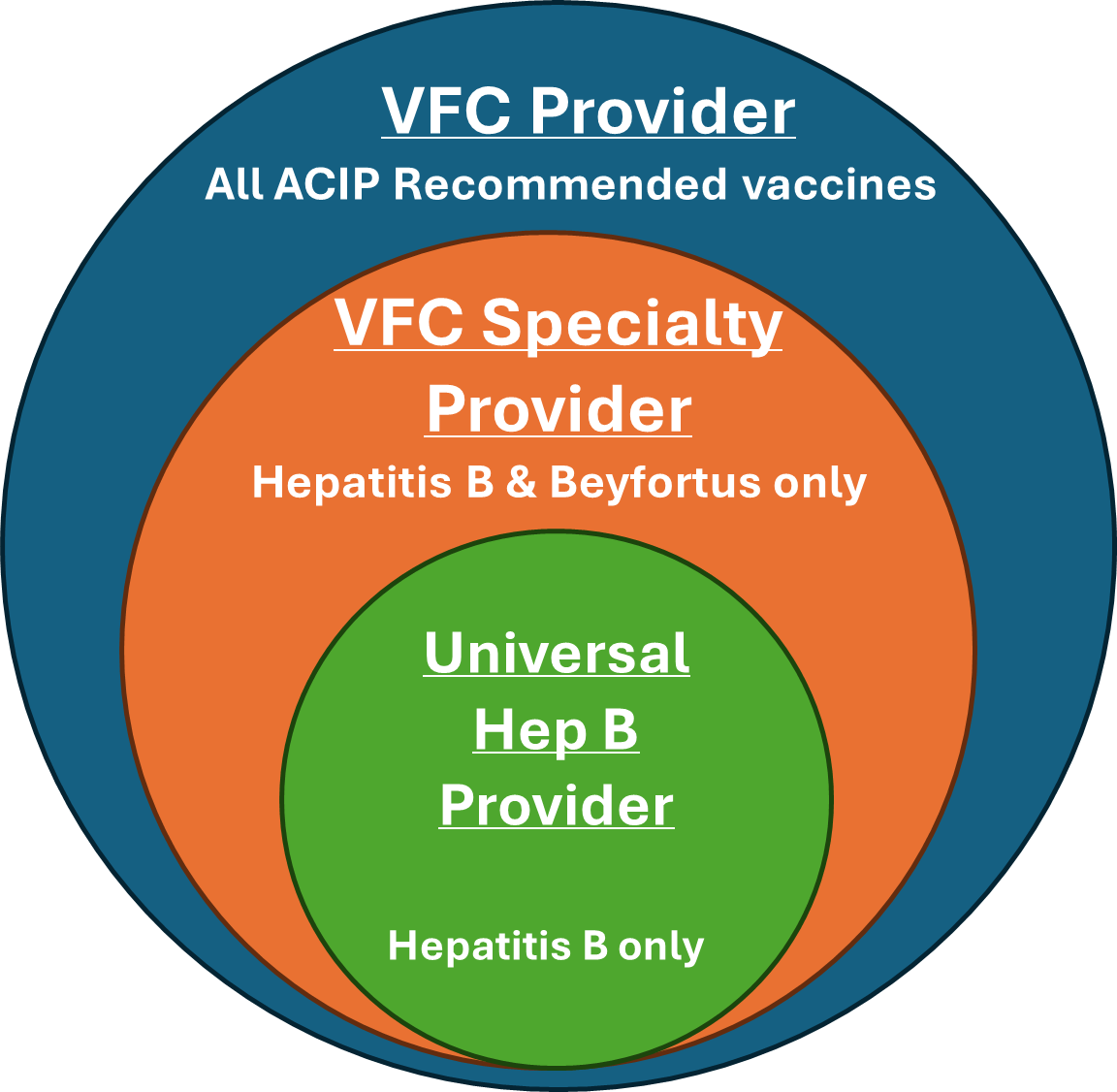
The Centers for Disease Control and Prevention (CDC) recently updated its guidance for the COVID-19 and varicella (chickenpox) vaccines. The CDC’s decision uses an individual-based decision-making framework, referring to vaccination decisions made through shared clinical …
MHA Endorsed Business Partner AMN Healthcare recently released the Rural Physician Recruiting Challenges and Solutions white paper, produced by its Physician Solutions division (formerly Merritt Hawkins). The resource identifies several approaches to address these challenges: Recruiting physicians …
When the One Big Beautiful Bill Act was signed into law in July, it created the Rural Health Transformation Program, a five-year, $50 billion investment in rural healthcare. The MHA Board of Trustees took swift action empowering an MHA board-appointed task force charged with creating …
Keckley Report

Shutdown Impact: The Affordable Care Act 2.0 Takes Center Stage
“In 2009, I facilitated discussions with key health industry trade associations and the White House Office of Health Reform focused on reforms to reduce costs, increase insurance coverage and improve quality by 2019. It was the Obama administration’s aim to use the health system’s bulk as a lever to stimulate recovery from the 2008-2009 recession and simultaneously increase coverage through Medicaid expansion and marketplace subsidies that for lower-income households. …
The current federal government shutdown is a tipping point for healthcare in the U.S. It’s about more than extended subsidies per Dem’s and holding the line on spending per Republicans. It’s about a growing sense of helplessness among the majority and resentment among many that institutions like the federal government, higher education, big business and healthcare are no longer motivated to serve interests beyond themselves. …
Some will harken back to the Affordable Care Act in 2010 when coverage was also the issue. We’re there again. But the bigger issue is this: extending subsidies and maintaining coverage will not lower spending or transform U.S. healthcare to an affordable, accessible, appropriately structured system of health.
The moral high ground for healthcare is in jeopardy and its direction unclear. Perhaps PPACA 2.0 is an answer. Doing nothing isn’t.”
Paul Keckley, Oct. 5, 2025